OVERVIEW
The South African National Cancer Registry (NCR), is a division of National Health Laboratory Services (NHLS). The NCR serves as South Africa’s main source of national cancer incidence data. Since 1986, the NCR has conducted pathology-based cancer surveillance, where data on cancer cases diagnosed by histology, cytology and bone marrow aspirate and trephine are collected, analysed and reported annually.
In 2011, Regulation no. 380 of the National Health Act no. 61 of 2003 was created to legally establish the NCR and to make cancer a reportable disease. The regulation provided a mandate to establish a population-based cancer registry. Since January 2017, population based cancer data are collected from Ekurhuleni metropolitan municipality in Gauteng Province. The Ekurhuleni Population Based Cancer Registry (EPBCR) meets the gold standard criteria for cancer registration worldwide as all cancers that are diagnosed clinically, radiologically and pathologically are collected.
CANCER REGISTRY
Healthcare Workers – Early Detection of Childhood Cancers
OBJECTIVES
The objectives of NCR are:
- To collect, analyse and report cancer statistics in South Africa;
- To guide and support the Department of Health on cancer initiatives and other cancer programs from NGOs, NPOs and any other parties interested in cancer prevention, control and care in South Africa; and
- To inform cancer policy and guideline in South Africa.
Functions
The South African National Cancer Registry (NCR) has been conducting national pathology-based cancer registration since its establishment in 1986. Data (demographic, clinical and reporting source) on cancer cases diagnosed either by histology, cytology, bone marrow aspirate and or trephine are submitted to NCR from both private and public laboratories across South Africa. The collected data is cleaned, analysed and reported in the annual cancer incidence report by sex, age and population groups. The national pathology-based cancer registry has been the primary source of cancer statistics for the country and has played a critical role in the development of cancer prevention and control policy for the country.
Methodology
The national pathology-based cancer registry methodology follows that of the World Health Organisation (WHO) International Agency for Research on Cancer
CANCER REGISTRATION: PRINCIPLES AND METHODS
Data sources
Data sources include National Health Laboratory Services (NHLS), Ampath, Gritzman & Thatcher, Lancet, Vermaak and partners.
Data management flow
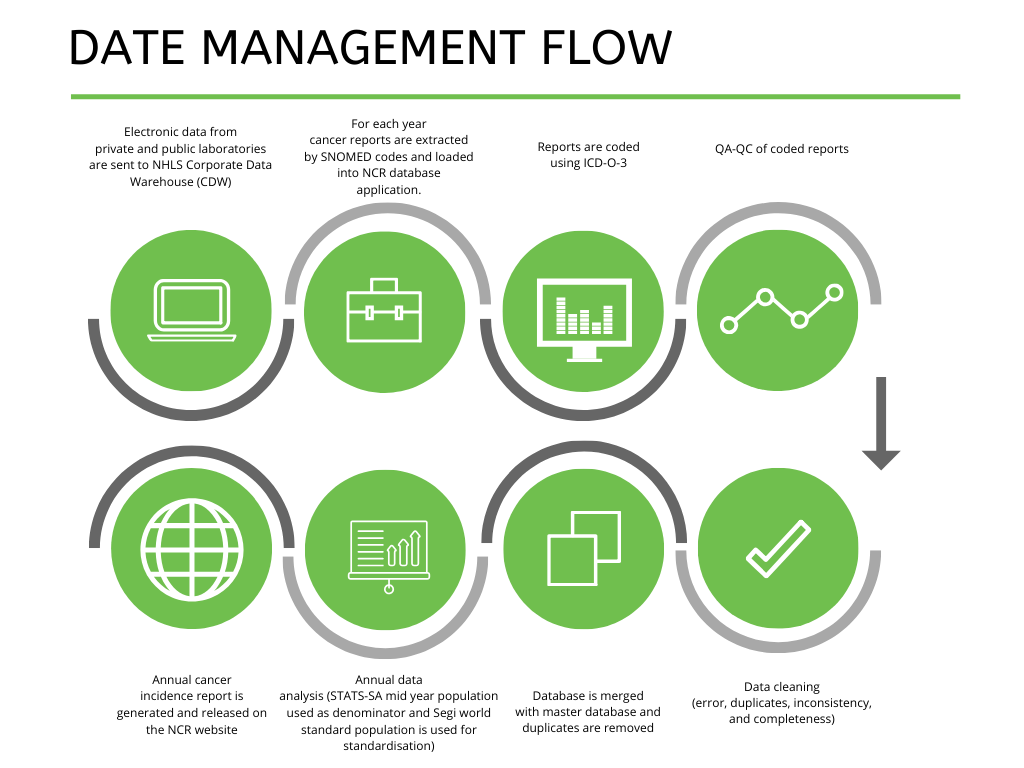
The Ekurhuleni population-based cancer registry (EPBCR) is a population-based cancer surveillance system that collects, analyses and reports all malignant cancers for individuals residing in the Ekurhuleni Metropolitan Municipality, Gauteng province of South Africa. The EPBCR was established in 2017 under the mandate of regulation no.380 of the National Health Act of 2003, access the National Health below.
Methodology
The registry uses both active and passive methods of data collection, and the processes of data collection follow the rules and standards recommended by the International Agency for Research on Cancer, see Cancer Registration Principles here.
The EPBCR registrars also known as surveillance officers (SOs) use data-source tools such as medical files, pathology results, pharmacy prescription registers, cancer screening registers (e.g. Pap smear registers), hospital admission registers, hospice registers, private practitioners consultation registers/records, radiological registers (e.g. CT scan and X-rays), discharge registers, mortuary registers and outpatient registers to identify, trace and notify Ekurhuleni cancer patients.
The SOs use the Republic of South Africa Department of Health’s cancer registration form to notify only Ekurhuleni residents’ cancer diagnosis. The International Classification of Diseases for Oncology (ICDO-3) is used as a reference for the coding of neoplasms. Completed cancer notification forms are either electronically captured remotely at surveillance sites by the SOs or centrally at the National Cancer Registry (NCR) by dedicated data capturers. The data capturing is done through the use of the Research Electronic Data Capture (REDCap) database system. In addition to the notification forms, the South African Oncology Consortium (SAOC) and the South African paediatric oncology network submit electronic cancer data in the form of excel spreadsheets or SQL database exports to the NCR at regular intervals. The electronic data are coded and incorporated into the REDCap database.
Furthermore, the national pathology-based cancer registry data for Ekurhuleni Metropolitan Municipality is extracted for the year of surveillance and linked to REDCap database to identify cases that are missed by the SOs, SAOC and the South African paediatric oncology network. The list of missed cases is then provided to the SOs to trace the missing cases back to medical records at the relevant health facilities. Once the missing cases are identified, the medical records and the pathology records are consolidated and captured. When case notification for a particular year is completed, the REDCap database for that year will be closed for data quality control and data quality assurance workflows. All malignant cases (M-80003-99893) from all body sites (C00.0-C80.9), excluding non-melanoma skin cancer (C44), are exported into Stata® statistical software (StataCorp LLC, Texas, USA) for cleaning and analysis. The annual report is then drafted and released, reporting cancer counts, frequencies, crude rates, lifetime risks and age-standardised incidence rates by sex and population groups for each cancer and a total thereof. The Segi world standard population is used for standardisation and mid-year population estimates from Statistics South Africa (STATS SA) are used as the population denominator.
Data use and permission
Careful consideration needs to be taken when using the data for EPBCR. The statistics provided here are that of Ekurhuleni population only and are not to be confused with the statistics of the national pathology-based cancer registry.
For permission to use NCR data, contact Dr Mazvita Muchengeti @ MazvitaM@nicd.ac.za
Annual Report
EKURHULENI POPULATION-BASED CANCER REGISTRY ANNUAL REPORT (2022)
EKURHULENI POPULATION-BASED CANCER REGISTRY ANNUAL REPORT (2021)
EKURHULENI POPULATION-BASED CANCER REGISTRY ANNUAL REPORT (2020)
EKURHULENI POPULATION-BASED CANCER REGISTRY ANNUAL REPORT (2018)
EKURHULENI POPULATION-BASED CANCER REGISTRY ANNUAL REPORT (2017)
Childhood Cancers
Currently, the National Cancer Registry includes all reported cases of childhood cancers (0-14years) within the annual pathology-based cancer incidence report. However, given the impact of childhood cancer on patients, families and society, the National Cancer Registry is working towards establishing a dedicated childhood cancer registry using the International Classification of Childhood Cancers (ICCC-3), as recommended by the International Agency for Research on Cancer (IARC).
This childhood cancer registry will provide a more complete and updated estimation of the burden of childhood cancer in South Africa. By publishing annual childhood cancer incidence reports, the NCR will increase awareness of childhood cancers amongst the general public, and the medical community so that there is a higher index of suspicion leading to earlier diagnosis and improved outcomes.
New cancer cases will be identified from the existing pathology-based registry, the Ekurhuleni population-based registry and notifications from clinicians.
Annual Report
CHILDHOOD CANCER REGISTRY REPORT (2020)
CHILDHOOD CANCER REGISTRY REPORT (2019)
CHILDHOOD CANCER REGISTRY REPORT (2018)
Notification Of Childhood Cancers
To report all new paediatric cancer cases to the NCR, as mandated by the No 380 regulation, please fill in the notification form and submit to cancer.registry@nhls.ac.za
Who Should Report?
As per No 380 regulation:
- Clinicians including Paediatric Oncologists and Clinical Haematologists
Any clinician who diagnoses a new case of cancer in a child under the age of 15 should submit a notification form within three months of diagnosis.
- Health establishments
The person in charge of a health establishment (including clinicians/nurses), where a diagnosis is confirmed must submit a notification form within three months of diagnosis.
- Laboratories
The head of a laboratory, on a quarterly basis, must ensure that notification forms, as well as the accompanying laboratory reports, are completed and submitted to the NCR.
For further information or queries, please contact the NCR administrator, Ms Evelyn Nkosi, at EvelynN@nicd.ac.za or 011-555-0548.
- The South African HIV Cancer Match Study (SAM)
The National Health Laboratory Services (NHLS) laboratories serve over 80% of the South African population and provide the unique opportunity to link longitudinal laboratory HIV data from the NHLS to cancer data from the NCR. This allows for the assessment of cancer risk in HIV-positive people in the era of antiretroviral treatment (ART) availability in South Africa, at national level.
- The Johannesburg Cancer Study (JCS):
The aims of the Johannesburg Cancer Study are to determine whether risk factors identified for cancers in developed countries apply to Black (African) adult patients attending tertiary public hospitals in Johannesburg, South Africa, and to understand the impact of HIV on cancer risk, with a view to identifying previously unrecognised HIV associated cancers (which may themselves, have an underlying infectious cause).
3. Evolving Risk Factors for Cancers in African Populations (ERICA-SA) study:
Lifestyle, infection, genetic susceptibility and cancer in South Africa: development of research capacity and an evidence base for cancer control.
4. Men of African Descent and Carcinoma of the Prostate Network (MADCaP):
Cancer of the prostate (CaP) is the leading cancer among men in sub-Saharan Africa (SSA). A substantial proportion of these men with CaP are diagnosed at late (usually incurable) stages, yet little is known about the aetiology of CaP in SSA.
LEADERSHIP AND TEAM

Dr Mazvita Muchengeti is a cancer epidemiologist who oversees cancer surveillance and research at the National Cancer Registry (NCR). Her interests lie in strengthening cancer surveillance and research in South Africa and the sub-Saharan African region, while exploring the role of infections in cancer development in African populations. She holds a medical degree from the University of Zimbabwe, a Diploma in HIV Management from the College of Medicine of South Africa, a master’s degree in Epidemiology and Biostatistics from the University of the Witwatersrand and a PhD from the University of Bern, Switzerland (2016).
Her doctoral work explored the spectrum and incidence of HIV-associated cancers in South Africa. Building on this work, she collaborates with the IeDEA Southern Africa, ISPM Bern and the Swiss TPH in the ongoing surveillance of HIV-associated cancers in South Africa through the South African HIV Cancer Match Study (SAM). This is a national cohort of HIV-positive adults and children linked to the NCR to explore the epidemiology of HIV-associated cancers at national level, in the context of the evolving HIV epidemic.
Muchengeti is a recipient of several awards, including the William Harding Le Riche Medal of Epidemiology, the Prestigious Postgraduate Degree award, the CRDF Global Beginner Investigator Grant for Catalytic Research in Cancer, the Africa-Oxford Initiative Travel Grant to study cervical cancer survival in sub-Saharan Africa and the Africa-Oxford Research Development Award to strengthen Childhood Cancer Surveillance in Southern Africa. She is also a recipient of the African Cancer Leaders Institute award with the responsibility of initiating, networking and leading research in cancer epidemiology in African populations.
She is an active member of the African Cancer Registry Network, supporting the network in training and grant applications. As an honorary senior lecturer at the University of the Witwatersrand’s School of Public Health, Epidemiology and Biostatistics Division, she lectures and supervises post-graduate epidemiology students and is a research associate at the South African Centre for Epidemiological Modelling and Analysis (SACEMA). She also supports the South African Field Epidemiology Training Programme through the supervision of SAFETP residents.
Administrator
Ms Evelyn Nkosi
Tel: +27 11 555 0548
Email: evelynn@nicd.ac.za
CANCER STATISTICS
The systematic collection, storage, analysis, interpretation and reporting of cancer cases known as cancer surveillance, is the responsibility of the National Cancer Registry (NCR) in South Africa.
The NCR is mandated to establish Population-Based Cancer Registries for South Africa. The findings are published annually, after undergoing stringent vetting processes. We at the NICD support the work of the NCR and host each year’s findings on our site.
Data sources that are included in the NCR’s research are among the likes of public hospitals, private health facilities, the South African Oncology Consortium, Hospice, paediatric oncology, CHOC, and the NHLS. A list of support contacts is provided below, and the NICD encourages you to contact the service providers if you are in need of oncology-related support.
























