1. Why should I get vaccinated if the vaccine already needs a booster shot to protect me? Are vaccines not effective?
Vaccines have been shown in controlled trials and in the evaluation of national vaccination programmes (e.g. United Kingdom) to reduce severe disease and mortality by up to 95-97%. This lifesaving effect has continued, even as variants have emerged. Taking a vaccine will protect you and your loved ones from a devastating illness and/or death. Recently, vaccines have also been shown to reduce transmission of COVID-19 from one person to another. As early as the first clinical trials, vaccines have been shown to reduce but not prevent mild to moderate illness.
As COVID variants has emerged, laboratory research has shown that protection against COVID-19 is related to antibody levels. Booster vaccination with the JnJ or Pfizer vaccine is a reliable and safe way of increasing antibody levels.
2. Why are booster shots recommended?
Booster vaccination increases antibody levels and ‘T-cell responses’ to SARS-CoV-2. Laboratory testing has shown that high antibody levels are more effective at neutralising variants of SARS-CoV-2. Therefore booster vaccines are helpful to improve protection from infection, especially during the fourth wave which is predominantly due to the Omicron variant.
3. Are booster shots safe?
Side effects of booster vaccines are similar to side effects from a first vaccine. Common side effects include tiredness, body aches, and pains, low-grade fever, pain at the injection site. Persons who have a booster vaccine may experience these common side effects more frequently. These side effects resolve completely within 24-48 hours.
Occasionally, very rare side effects (for example, myocarditis, which may occur at 12.6 cases/million doses of Pfizer vaccine) may occur slightly more frequently after a second (or booster) vaccine. If you have experienced any side effects after your first vaccine, you should report the side effect on the MedSafety app (see https://aefi-reporting.sahpra.org.za/report-aefi.html) , and discuss the need for a booster with your health care provider.
4. Who should get a booster shot, and when?
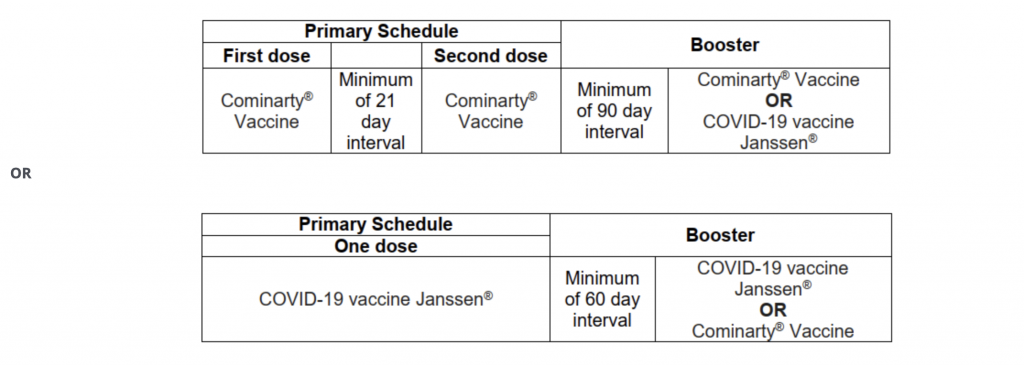
Cominarty® (Pfizer) vaccine:
- From 23 February 2022, individuals older than 18 years of age who received their first dose of the Pfizer vaccine can get their second dose after 21 days (previously, this was 42 days).
- Individuals older than 18 years of age who received their second dose of the Pfizer vaccine can get their third booster dose after 90 days (previously this was 180 days).
Johnson & Johnson vaccine:
- From 21 February 2022, individuals older than 18 years of age who received their first dose of the J&J vaccine can get their second booster dose after 60 days.
5. Can you mix and match vaccines?
You are now able to get a mixture of the J&J and Pfizer vaccines against Covid-19 in South Africa. You just have to be fully vaccinated with the one, then ask for the other as a booster, after either 60 or 90 days, depending on which you got first.
6. Which booster shot will be administered?
The decision regarding which vaccine to administer as a booster should be guided by vaccine availability. If both vaccines are available, homologous boosting should be preferred, unless the vaccine requests to receive a heterologous booster dose, or has a history of experiencing an adverse event following immunisation. (Please remember that all adverse events following immunisation must be reported).
7. Will a booster help against omicron?
Yes. There is good evidence that booster vaccination increases antibody levels to the spike protein of the SARS-CoV-2 virus and that higher antibody levels offer more protection against variants, including Omicron.
8. What if I’m immunocompromised, does the timing of the third shot differ?
Persons who are immunocompromised should receive an additional Pfizer or Johnson and Johnson vaccine dose at least 28 days after receiving their last dose. Persons who are immunocompromised should be referred by their doctor for a booster dose
Presently, persons are considered immunocompromised if they have
- Haematological or immune malignancy
- Moderate to Severe Primary immunodeficiency disorder
- HIV infection with CD4 count < 200 cells/µL within the last 6 months
- Asplenia
- Individuals receiving the following treatments:
- High dose steroids or systemic biologics (e.g. for autoimmune conditions)
- Long term renal dialysis
- Transplant recipients (Solid organ or bone marrow)
The additional vaccine dose should be of the same vaccine type as the initial dose (or doses).